Fragile X-associated Primary Ovarian Insufficiency (FXPOI)
| Prevalence |  Fragile X syndrome, the most common heritable form of mental retardation in males, affects ~1/4000 males[1]. It occurs in most ethnic and racial populations, but incidence may vary from group to group[2]. Fragile X syndrome, the most common heritable form of mental retardation in males, affects ~1/4000 males[1]. It occurs in most ethnic and racial populations, but incidence may vary from group to group[2].
Fragile X syndrome is an X-linked genetic disorder caused by a mutation in the FMR1 gene. Female carriers may pass it on to their children. Male children are expected to have the physical and behavioral features of the condition. Some females with one copy of the mutation have been reported to have features of the condition, but with lower frequency and milder involvement. Women who carry a Fragile X premutation are at increased risk to develop Fragile X-associated primary ovarian insufficiency (FXPOI) and Fragile X-associated tremor/ataxia syndrome (FXTAS). In the largest US study to date (>119,000 women), the overall carrier frequency of Fragile X among US females was found to be 1.3%[3]. Among women with primary ovarian insufficiency (POI), between 4-6% are Fragile X carriers[4]. |
| Natural History | The FMR1 gene, located on the X chromosome, was identified in 1991 and was found to include a CGG trinucleotide repeat within the first exon (coding region). Expansion of this trinucleotide repeat leads to a group of disorders: Fragile X, Fragile X-associated primary ovarian insufficiency (FXPOI), and Fragile X-associated tremor/ataxia syndrome (FXTAS). The size of the repeat correlates with the disorders an individual is at risk to develop:
The distinction between intermediate and premutation alleles is made by family history and repeat instability. Premutation alleles are unstable and can expand to a full mutation (>200 repeats) is some offspring. In contrast, intermediate alleles are not known to expand to full mutations, therefore offspring are not at risk for Fragile X syndrome. However, approximately 14% of intermediate alleles are unstable and may expand into the premutation range when transmitted by the mother[6]. To date, no allele 56 repeats or smaller has been known to expand to a full mutation in 1 generation[7]. It is important to note that the presence of AGG interruptions within the CGG trinucleotide repeat reduces the risk of transmission of a full mutation for all maternal premutation repeat lengths below ~100 CGG repeats[8]. |
| Diagnosis | Fragile X syndrome carrier status is often diagnosed in these three ways:
|
| Disease Presentation | 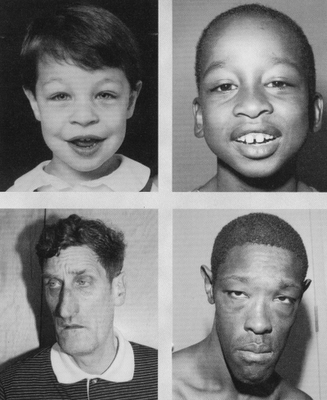 Fragile X Syndrome Fragile X Syndrome
Fragile X syndrome is characterized by mental retardation or impairment which ranges from borderline, incuding learning disabilities, to severe. It also presents with behavioral disabilities, including autism[5,9,10]. Phenotypic abnormalities include: distinctive facial features (long narrow face, prominent ears), macroorchidism, connective tissue problems, and speech and language difficulties[9]. Fragile X-associated tremor/ataxia syndrome (FXTAS) Male and female FMR1 premutation carriers are at increased risk to develop FXTAS, a late-onset neurodegenerative disorder characterized by progressive cerebellar ataxia, intention tremor, memory loss, anxiety, reclusive behavior, and dementia. Onset is typically after age 50 years and is more common in male carriers than female. Risk and severity are directly related to the size of the premutation repeat[5,11]. Fragile X-associated primary ovarian insuffieciency (FXPOI) Women with FMR1 premutations are at a ~10-fold increased risk (~20%) of developing premature ovarian insufficiency (POI) or premature ovarian failure (POF)- diagnosed by amenorrhea and postmenopausal levels of follicle stimulating hormone (FSH) before the age of 40[12,13]. The risk of POI is dependent on the number of CGG repeats, with peak risk of POI at 80-100 repeats[14]. Women with full mutation alleles are not at increased risk for POI. |
| Effect on Fertility | Approximately 20% of female FMR1 premutation carriers develop POI, defined as cessation of menses before age 40 years[13]. Of those who develop FXPOI, 1/3 will experience final cessation of menses at or before age 29 years and 1% will have cessation of menses prior to age 18[14]. In fact, ~3% of adolecents with FMR1 premutations will experience some menstrual cycle irregularity[14]. |
| Fertility Preservation Options | Early in reproductive life, all FMR1 premutation carriers should be provided with information regarding the risk of developing FXPOI and presented with fertility preservation options to consider.
Currently, there are no successful therapies to regain ovarian function in women with POI. There are, however, hormone replacement therapy (HRT) and fertility preservation options available to women at increased risk for and with POI. Hormone Replacement Therapy Estrogen replacement therapy is recommended for women with POI by the American Society for Reproductive Medicine and the International Menopause Society. Hormone therapy in adolescent girls and young women with estrogen and progestin replaces what the ovary should have naturally produced at that age. The exogenous hormones will also promote the development of secondary sexual characteristics in those who have failed to naturally develop these characteristics. Adolescent girls and young women would be at low risk for cardiovascular complications from estrogen and progestin, however there is no long term data to delinate the risk-benefit profile of hormone therapy for girls with POI[15]. Fertility Preservation Women with FMR1 premutations and family history of POI may be interested in fertility preservation options, including ooctye, embryo, and ovarian tissue cryopreservation.
Ovarian Tissue Cryopreservation *It is important to not assume infertility in women with FXPOI. There is a 5-10% chance of pregnancy in the POI general population, it is unknown if this applies to the FXPOI population as well[16]. Women with FXPOI who are not trying to conceive should use contraceptives. |
| Additional Pregnancy Concerns | While premutation carriers do not have Fragile X syndrome, they can pass on a full mutation to their children. The likelihood of a carrier mother passing the full mutation to her children increases with the size of her premutation repeat allele, especially if she has at least 100 CGG repeats in the FMR1 gene[6]. It is recommended that women with FMR1 premutations (as well as intermediate mutations and full mutations) receive genetic counseling to discuss the availability of the option of preimplantation genetic diagnosis (PGD) or prenatal testing (amniocentesis and chorionic villus sampling (CVS)) during pregnancy. |
| Emotional Well-Being | The diagnosis of POI can be devastating to young women who may not have completed, or even started, family planning. It is well established that loss of fertility can lead to emotional distress and women with FMR1 premutations may be at increased risk for depression and anxiety[17]. It is recommended that women with FMR1 premutations receive a follow-up visit to screen for symptoms of depression and anxiety. |
| Fragile X Carrier Testing | The American College of Medical Genetics (ACMG) has issued a policy statement recommending fragile X testing for “women with reproductive or fertility problems associated with elevated FSH levels, especially if there is a family history of premature ovarian failure, fragile X syndrome, or undiagnosed mental retardation”[5,18]. The Genetics Committee of the American College of Obstetrics & Gynecology suppports the ACMG recommendation[9]. |
| Patient Fact Sheet | Print the Oncofertility Consortium® Fertility Preservation Patient Fact Sheet on Fragile X-associated Primary Ovarian Insufficiency (FXPOI) to provide to your patients and their families who are considering fertility preservation due to premature ovarian insufficiency. This fact sheet is tailored specifically to the fertility needs of patients and families with FXPOI.
Fragile X-associated Primary Ovarian Insufficiency (FXPOI) Fertility Preservation Patient Fact Sheet |
References
[1]Crawford, D., Acuna, J., & Sherman, S. (2001). FMR1 and the fragile X syndrome: human genome epidemiology review. Genet Med, 3, 359-371.
[2]Crawford, D., Meadows, K., Newman, J., Taft, L., Scott, E., Leslie, M., . . . Sherman, S. (2002). Prevalence of the fragile X syndrome in African-Americans. Am J Med Genet, 110, 226-233.
[3]Strom, C., Crossley, B., Redman, J., Buller, A., Quan, F., Peng, M., . . . Sun, W. (2007). Molecular testing for Fragile X Syndrome: lessons learned from 119,232 tests performed in a clinical laboratory. Genet Med, 9, 46-51.
[4]Sullivan, S., Welt, C., & Sherman, S. (2011). FMR1 and the continuum of primary ovarian insufficiency. Semin Reprod Med, 29, 299-307.
[5]Monaghan, K. G., Lyon, E., Spector, E. B., & American College of Medical Genetics and Genomics. (2013). ACMG Standards and Guidelines for fragile X testing: a revision to the disease-specific supplements to the Standards and Guidelines for Clinical Genetics Laboratories of the American College of Medical Genetics and Genomics. Genet Med, 15(7), 575-586.
[6]Nolin SL, Glicksman A, Ding X, Ersalesi N, Brown WT, Sherman SL, Dobkin C. (2011). Fragile X analysis of 1112 prenatal samples from 1991 to 2010. Prenat Diagn, 31:925–31.
[7]Fernandez-Carvajal I, Lopez Posadas B, Pan R, Raske C, Hagerman PJ, Tassone F. (2009). Expansion of an FMR1 grey-zone allele to a full mutation in two generations. J Mol Diagn,11:306–10.
[8]Yrigollen, C., Durbin-Johnson, B., Gane, L., Nelson, D., Hagerman, R., Hagerman, P., & Tassone, F. (2012). AGG interruptions within the maternal FMR1 gene reduce the risk of offspring with fragile X syndrome. Genet Med, 14(8), 729-736.
[9]American College of Obstetrics and Gynecologists. (2010). Carrier Screening for Fragile X Syndrome. Committee Opinion No. 469. Obstet. Gynecol., 116, 1008-1010
[10]Pastore, L., Morris, W., & Karns, L. (2008). Emotional Reaction to Fragile X Premuation Carrier Tests Among Infertile Women. J Genet Couns, 17(1), 84-91.
[11]Hagerman, P., & Hagerman, R. (2004). The fragile-X premutation: A maturing perspective. Am J Hum Genet, 74(5), 805-816.
[12]Sullivan, A., Marcus, M., Epstein, M., Allen, E., Anido, A., Paquin, J., . . . Sherman, S. (2005). Association of FMR1 repeat size with ovarian dysfunction. Hum Reprod, 20(2), 402-412.
[13]Ennis, S., Ward, D., & Murray, A. (2006). Nonlinear association between CGG repeat number and age of menopause in FMR1 premutation carriers. Eur J Hum Genet, 14, 153-155.
[14]De Caro, J., Dominguez, C., & Sherman, S. (2008). Reproductive health of adolescent girls who carry the FMR1 premutation: expected phenotype based on current knowledge of fragile X-associated primary ovarian insufficiency. Ann N Y Acad Sci, 1135, 99-111.
[15]Baker, V. (2013). Primary ovarian insufficiency in the adolescent. Curr Opin Obstet Gynecol, 25(5), 375-381.
[16]Nelson LM, Covington SN, Rebar RW. (2005). An update: spontaneous premature ovarian failure is not an early menopause. Fertil Steril, 83:1327–32.
[17]Nelson, L. (2009). Clinical practice. Primary ovarian insufficiency. N Engl J Med, 360(6), 606-614.
[18]Sherman, S., Pletcher, B., & Driscoll, D. (2005). Fragile X syndrome: Diagnostic and carrier testing. Genet Med, 7(8), 584-587.
[19]Finucane, B. , Abrams, L., Cronister, A., Archibald, A.D., Bennett, R.L., McConkie-Rosell, A. (2012). J Genet Couns, 21(6): 752-760.
About the Author
Allison Goetsch, MS, CGC is a pediatric genetic counselor at Ann and Robert H. Lurie Children’s Hospital of Chicago and a member of the Oncofertility Consortium administrative core team. She completed her graduate thesis with Dr. Teresa K. Woodruff researching oncofertility and hereditary breast and ovarian cancer (HBOC) syndrome. Allison’s primary goals are to increase fertility preservation awareness and education for both health care providers and patients regarding malignant and non-malignant diseases (and/or treatments) which threaten fertility.
This page was last updated February 6th, 2016.